Peptide Synthesis: From Research Tools to Clinical Applications
Jan 16th 2024
Recent biotechnological breakthroughs have expanded the horizons and capabilities of today's biochemists, ushering in an era of custom compound creation. Where previous generations synthesized natural product derivatives through iterative trial-and-error, contemporary scientists now engineer bespoke antibodies, proteins, peptides, and beyond - crafted to client specifications rather than arising serendipitously.
Based on these innovations, improved purification techniques guarantee unprecedented quality and consistent results for commissioned biological materials. Once sold in crude isolates under harsh reaction conditions, the same compounds achieve clinical and research-grade purity through technological might.
Peptide synthesis represents one key area experiencing enhancements through the attendant upgrades in amino acid coupling chemistries, automation, and analysis methods. Where peptide creation previously existed as a cumbersome practice, the collective innovations now make custom concatenation of amino acid sequences routine. Despite the sea changes in achievable lengths and processing ease, much knowledge about peptide synthesis remains guarded within the industry.
Please read the following blog to understand peptide synthesis and its applications in the modern biochemical field.
What is Peptide Synthesis?
Living cells harbor innate peptide synthesis machinery inside ribosomes, so nature-imposed length limits cannot fulfill scientific and therapeutic demands for custom lengths. Myriad research and clinical applications require peptides not naturally encoded by an organism's genome.
Chemists developed bespoke peptide synthesis protocols to transcend biological barriers to construct novel chains expanding beyond endogenous constraints. Chiefly, solid-phase peptide synthesis (SPPS) revolutionized the field by automating amino acid coupling reactions on inert resin supports. In contrast to solution-based procedures, solid-phase synthesis permits rapid assembly of specified sequences without length restrictions through repeating coupling baths.
Mirroring DNA synthesizers, automated SPPS integration now allows commercial providers to deliver high-throughputpeptide synthesis services, keeping pace with orders from diverse research institutions and industry verticals.
Custom projects, once requiring months of toil, are now completed in weeks with purity and accuracy certification. Some synthesis partnerships extend custom peptide services into complementary domains like antibodies, proteins, and gene fragments.
What are The Techniques Applied in Peptide Synthesis
1. Solid-phase peptide synthesis (SPPS)
Among the peptide synthesis methods, solid-phase peptide synthesis (SPPS) reigns supreme for efficient concatenation of amino acid chains. Supported by insoluble beaded resin substrates, SPPS leverages successive reactive baths of amino acid derivatives to construct polypeptides rapidly through repeating deprotection and coupling cycles. Whereas previous practices demanded intricate enzyme-mediated synthesis or laborious liquid-phase procedures, SPPS instead works its magic automatically from the attachment of the resin bead's first amino acid.
By transitioning the growing peptide chain between microreactors, SPPS enables the mass production of high-purity peptides without immense chemist intervention. The technological advancement catalyzed a seismic shift in research and therapeutic development previously hindered by throughput bottlenecks. Now empowered to produce peptides at scale reliably, discovery programs expedited the unveiling of myriad bioactive agents.
And the innovations march forward - where once stood days of crystallization protocols now simplify basic filtrations to collect the resin-borne cargo. As pharmaceutical interests continue to bankroll enhancements around coupling reagents and analytical methods, SPPS promises to accelerate customized peptide synthesis from minute research quantities to metric tons.
2. Classical solid-phase synthesis (SPS)
Pre-dating contemporary solid-phase peptide synthesis (SPPS), classical solid-phase synthesis (SPS) pioneered resin-anchored concatenation of amino acids, though with more excellent chemist input required per coupling. Rather than automating the assembly of chains by progressively tunneling down sequence lattices like in SPPS, SPS instead binds monomers one by one in iterative baths. While demanding extensive precipitation and purification steps post-synthesis, SPS allows certified intermediate conformation between each reaction cycle.
Befitting the analog era origins, SPS favors crafting longer peptides that are not as easily tamed by early methodologies. Chemists couple shorter peptide fragments first via SPS before fusing them in convergent synthesis, culminating in elongated sequences. The ability to immediately purify reaction batches before advancing enables certified buildings to move upward from verified bases.
Human insulin constituted one early SPS success alongside other hypothalamic, gastrointestinal, and glandular peptides. Without SPS laying conceptual foundations, SPPS may have lacked springboards for automation. However, the manual workflow imparts lengthy synthesizing timelines. Streamlining arrived through enhanced coupling agents and resin-anchored strategies ushered by SPPS in response to asynchronous demands. Yet for bespoke peptides beyond current machine limits, SPS persists as a unique means for accessing exotic lengths.
3. Peptide Purification
While representing the culmination of peptide synthesis endeavors, purification ensures reaction completeness by removing residual reagents, truncated sequences, and other impurities that accumulate throughout production. As the final guardian of quality, rigorously purifying crude peptide preps begets precision and consistency crucial for analytical and therapeutic applications.
The choice of purification methodology depends mainly on the upstream synthesis approach. For solid-phase methods, reversed-phase HPLC provides exceptional separative abilities to isolate target peptides from deletion sequences and scattering chemical remnants. Hydrophobic interaction methods may assist, while ion exchange and hydrophilic interaction chromatography offer selectivity by charge and polarity, respectively.
Tradeoffs exist, however - extensive solvent volumes concentrate environmentally while evaporative losses decrease yields. Repetitive chromatographic cycling also ratchets production timelines. Yet for certified standards and drug candidates, intensive purification bolsters quality assurances by extracting the peptide of interest away from obscuring contamination. Integrating analytical characterization like mass spectrometry and peptide mapping ensures a comprehensive purity portrait.
Here are The Major Applications of Peptide Synthesis
1. Production of Antibodies
Preventive medicine includes medical efforts to prevent illness, disability, or fatality. Such efforts either delay disease progression or prevent disease onset altogether. A significant aspect of preventive medicine is vaccine development and manufacturing; vaccines have helped wipe out infectious diseases like smallpox. Antibody engineering services and antibody analysis services are two critical components of preventive medicine.
As a result, commercial peptide production companies offer services to research institutions and the vaccine sector. Initially, antibody production involved injecting a host animal with an inactivated antigen to trigger an immune response and antibody production. After immunization, a serum collection process retrieved the antibodies from the host.
However, in vivo antibody production had several drawbacks, including the risk of antigen toxicity to researchers and immunogenicity in the host animal. Immunogenicity refers to the host's immune reaction to an antigen, which may cause an undesirable immune response. Fortunately, in vitro antibody production facilitated by peptide engineering is now possible, eliminating the need for animal hosts. Additionally, peptide production and antibody analysis services can now generate synthetic antibody libraries - databases of human-like synthetic antibodies. These antibody libraries also enable custom gene synthesis and custom antibody engineering.
Animal-free antibody production has multiple advantages over traditional and sequencing antibody production services. For instance, synthetic antibodies have higher affinity and specificity than animal-derived antibodies. It also cites faster generation and lower immunogenicity as benefits. Peptide production services for preventive medicine also providecustom antibody engineering. This involves creating tailored antibodies that may not exist naturally to meet specifications.
These antibodies primarily assist antibody research, including custom gene synthesis, protein production, and specialized purification. Bispecific antibodies are a significant advancement enabled by custom antibodies targeting two distinct antigens. Peptide antibodies developed through peptide research have also transformed preventive medicine, with some peptide vaccines targeting cancerous growths.
2. Production of Proteins
Proteins are cellular particles that control many vital body functions. Therefore, the high-volume creation of proteins for scientific and commercial purposes is made possible by recombinant protein engineering or protein synthesis services. Therefore, how is the synthesis of peptide services related to protein production? Amino acids are the building blocks of both peptides and proteins; peptides have shorter amino acid bounds, while proteins have lengthy ones. A protein molecule also comprises a sequence of polypeptide chains connected by amide bonds.
Furthermore, DNA determines the order of amino acids in peptides and protein chains. Thus, a firm that provides peptide synthesis probably alsofurnishes customized gene synthesis and protein production as part of an all-inclusive service plan. Gene synthesis involves generating and piecing together gene products such as DNA and proteins. Commercial peptide production companies generally make gene synthesis available to guide amino acid arrangement in peptides. This is critical because it gives peptides and proteins their intrinsic qualities and capabilities.
Historically, gene replication took place in living organisms, necessitating expression systems. However, progress in biotechnology enables gene synthesis providers to furnish services such as customized peptide synthesis to construct DNA devoid of a template. Moreover, DNA production enterprises do not constrain nucleotide length or base pair numbers in bespoke DNA synthesis. Additionally, peptide synthesis employs high-volume procedures to swiftly generate unfeasible top-notch polypeptide chains via old-fashioned protein expression services.
Personalized DNA synthesis made possible by customized gene synthesis offerings allows protein manufacturing providers to furnish bespoke protein production and synthesis solutions. These services create new proteins not naturally present for peptide and protein investigation. Gene synthesis firms possess gene collections for custom gene synthesis base pairs, expediting tailored DNA synthesis for personalized protein generation.
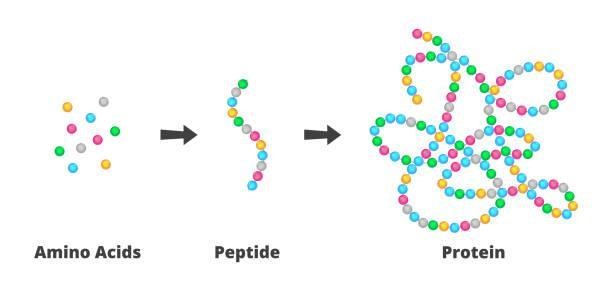
3. Peptide Therapeutic
Peptide therapeutics is a developing health field that employs peptides and polypeptides, or proteins, to cure a disease or regulate its signs. Insulin and ghrelin (hunger hormone) are some peptide therapeutic compounds used to manage blood glucose levels and increase appetite. DNA and gene synthesis companies lead the way in peptide therapeutics by making tailored peptide production and protein manufacturing possible. High-volume customized peptide synthesis and bespoke protein expression solutions aid exploration into the following peptide therapeutics areas:
Drug Development
Drug development harnesses a peptide's cellular capabilities to cure illness beyond the body's innate hormones. DNA and gene production companies assist peptide drug advancement and tailored DNA synthesis in creating novel or modified DNA varieties.
Custom protein fabrication has created peptide-based medications used as a pre-exposure prophylactic to prevent transmission of HIV. Reports indicate over 60 FDA-sanctioned peptide drugs as of 2018, with about 100 more candidates in various phases of clinical trials pending potential approval. These include peptide cancer medications from customized protein synthesis solutions.
Targeted Drug Transport
Targeted drug delivery amplifies a medication's concentration at a site, such as a cell or tissue, compared to the remainder of the body. As fundamental cellular components, peptides and polypeptides can be targeted drug transport carriers. Therefore, peptide manufacturing companies furnishing tailored protein expression and gene synthesis supply superior peptides and proteins for focused drug transport.
Regenerative Medicine
Regenerative medication fixes, regrows, or substitutes impaired tissue, organs, or cells to reinstate standard functionality. Though it chiefly utilizes stem cells, studies show pairing them with new peptides hastens regeneration. Protein manufacturing services providing gene synthesis can create gene arrangements for new peptides. Instead, regenerative medicine specialists can investigate gene collections at gene production firms for feasible novel gene sequences.
4. Medical Visualization
Medical visualization includes technologies like enzyme-linked immunosorbent assays (ELISA kits) and X-rays to see the body's musculoskeletal structure, interior tissues, and organs. It also comprises molecular imaging to remotely view, characterize, and quantify targeted molecules and cellular procedures. Radiolabeling or isotopic tagging is the most prevalent medical imaging approach. It necessitates binding a molecule to a radioactive material so the molecule becomes remotely detectable as it journeys between cells, tissues, and organs. Medical professionals and scientists leverage medical imaging to monitor functions like medication metabolism and disease pathogenesis for diagnosis.
Proteins and peptides are the most frequentlyutilized radiolabeled molecules in isotopic marking tagging owing to their strong binding affinity and selectivity to target cells. Furthermore, a protein generation service with protein expression and purification can alter the peptides and proteins to boost the radiolabeled protein's specificity and attraction during customized protein synthesis. In addition to remote observing, radiolabeled proteins and peptides are used by manufacturers of ELISA test kits in assay analyses to measure antigen density in biological materials. Also, test kit developers design varied ELISA test varieties enhanced for explicit assay roles. Furthermore, companies providing purification and protein expression services might create their isotopic tagging kit instead of pre-made kits from ELISA test suppliers.
5. Food Preservation
Large-scale food preservation depends on purification services and protein expression. As previously mentioned, many peptide and polypeptide types have distinct physiological roles. Antimicrobial or host defense peptides are an important class of peptides. They protect against pathogenic attacks and are an essential component of the immune system in humans and other creatures.
However, peptides with antimicrobial properties are utilized as food preservatives in manufactured and frozen foods. A study discovered that antimicrobial peptides exhibit stronger adhesion characteristics and reduced toxicity compared to chemical additions. Hence, they offer a natural, less hazardous substitute for traditional food preservative agents. Peptide production companies can manufacture antimicrobial peptides through recombinant techniques.
Wrapping Up
Peptide synthesis is vital in biochemistry, facilitating research in biological structures, enzyme inhibitors, and therapeutic agents. Technological advances have made peptide synthesis simple, fast, and customizable to meet research needs.
Peptide synthesis service costs vary depending on the desired sequence, production process, final peptide length, and purification methods. When seeking custom peptide services, opt for reliable manufacturers that are transparent about their process and components used.
Carefully reviewing this information can help determine if a provider offers the best quality custom peptide synthesis suited to your research aims and budget. Selecting the right custom peptide partner allows you to access a powerful tool for advancing projects across medicinal, therapeutic, and biochemical domains.